Green ammonia and how it relates to concentrated solar power
By Xavier Lara
Green ammonia: if you haven’t heard of it yet, then pay attention. It could play a key role in the energy transition. And it could be important for CSP in several ways. This is a big deal because ammonia today is the world’s second largest chemical commodity, used primarily to create fertilizer.
Global production capacity of the colorless, pungent gas is expected to expand from around 235 million metric tons in 2019 to almost 290 million metric tons by 2030. But its production today, via a chemical pathway called the Haber-Bosch process, is very carbon intensive.
The Haber-Bosch process uses fossil fuels—usually natural gas—as a fuel source and a feedstock, to an extent that in 2008 the industrial production of ammonia was said to represent 1% of global energy use. This amount doubled if production of hydrogen, an ammonia precursor, was taken into account.
That energy use translates to around 1% of global greenhouse gas emissions. On average, every ton of ammonia creates almost three tons of carbon dioxide. The question now being posed by scientists and industrial concerns is: can we produce ammonia without such a massive carbon footprint?
This is a pressing question not just because the demand for ammonia from the fertilizer industry is likely to grow, but also because the gas—if produced in a low-carbon fashion—is increasingly being seen as the simplest and lowest-cost way of decarbonizing the maritime transport sector.
That’s because it’s easier to store and transport ‘green’ ammonia than green hydrogen, the other main low-carbon fuel option. Volumetrically, a liter of liquid ammonia actually contains more hydrogen than a liter of liquid hydrogen, because of the makeup of the molecules.
Fortunately, it looks like low-carbon or green ammonia synthesis is indeed possible. And it may involve CSP in several ways. The first is to decarbonize production of hydrogen, one of the main ingredients in ammonia synthesis.

Like ammonia, hydrogen production currently relies on fossil fuels and is carbon intensive. Moves to produce low carbon ‘green hydrogen’ have focused on the electrolysis of water using low-cost electricity from wind or solar power. But CSP offers an alternative.1
Research almost a decade ago by the University of Colorado in Boulder showed how a solar receiver could power hydrogen synthesis from steam. The process would require a temperature of 1,350ºC but could achieve 20% efficiency, compared to 12% for PV to hydrogen via electrolysis.
And that’s just one line of research. Elsewhere, teams have also proposed two-step pyrolysis processes and reforming based on wastewater or agricultural crop residues, to cite just two approaches.
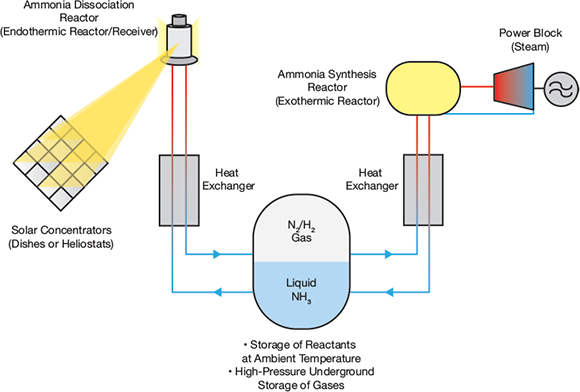
Beyond the production of green hydrogen, CSP could also be more closely linked to a low-carbon ammonia future. In one proposal, ammonia could substitute molten salt as an energy storage medium in CSP plants.
Researchers say this could significantly reduce the cost of CSP with storage, because ammonia could be stored in a single-tank arrangement on site and, for large volumes, underground in drilled shafts or salt caverns.
Using a salt cavern, researchers believe the installed cost of ammonia based CSP with six hours of storage could fall to as little as USD$13 per kWh (thermal).
This is within the US Department of Energy SunShot program goal of delivering a levelized cost of electricity of 6 cents per kilowatt-hour, based on an installed cost of thermal energy storage of $15 per kWh or less.
The use of ammonia as a storage medium takes advantage of a reversible Haber-Bosch process and so does not lead directly to the production of a low-carbon version of the gas. But it could avoid emissions by helping CSP plants deliver a greater share of dispatchable clean energy.
What if CSP could create green ammonia directly, though? This is the goal of the solar-thermal ammonia production (STAP) program being hatched at Sandia National Laboratories in the US.
The STAP team, which includes experts from the Georgia Institute of Technology and Arizona State University, notes that the Haber-Bosch process requires high pressures and temperatures of between 400ºC and 500ºC.
It has proposed a novel synthesis pathway whereby a solar thermochemical looping technology produces and stores nitrogen from air in order to produce ammonia. The inputs are sunlight, air and hydrogen, and the output is green ammonia.
The synthesis could be achieved a significantly lower pressures than the Haber-Bosch process. And by using green hydrogen it pretty much eliminates the carbon footprint of ammonia. The approach is still in its early phases, having been presented in 2019.
Getting to commercialization will require fine-tuning the synthesis and characterization of oxides for nitrogen recovery from air. Then it will need to look at the synthesis and characterization of nitrides for ammonia production.

That’s before it gets to the design, development and demonstration of processes and reactors. Finally, it will need to overcome rigorous system and technoeconomic analyses. But the prize is well worth the effort.
As mentioned above, ammonia is the world’s second most important industrial compound, and is set to grow in importance.
Decarbonizing ammonia production is therefore a key challenge in the fight against climate change and is not one that can easily be addressed through mainstream renewable energy technologies such as wind or PV.
Given the multiple ways in which CSP could help this process, and the fact that both ammonia production and solar thermal energy are mature technologies, it makes sense to invest in further research and development efforts in this area.
One stumbling block is that CSP is still perceived as a relatively expensive form of renewable energy generation, when in fact costs have fallen significantly even across a small number of recent projects. The reality is that CSP is capable of major cost reductions, particularly if deployed at scale.
And ammonia production offers a perfect opportunity for scaling, given the need to deliver millions of tons of product a year.
At Pacific Green Solar Technologies, we see green ammonia as a major opportunity for the business and are keen to work with investors and developers in driving forward new projects.
As recent interest in green hydrogen has shown, there is significant institutional appetite to embrace new low-carbon fuels. So let’s get going.